Annals of Burns and Fire Disasters - vol. X - n. 4 - December 1997
TRANSFER
OF AUTOLOGOUS KERATINOCYTES GROWN ON A POLYMER REDUCES TIME TAKEN FROM BIOPSY TO GRAFT
Burt
AM., Clarke J.A.
Stephen
Kirby Skin Bank, Burns Unif
Queen Mary's University Hospital, London, Great Britain
SUMMARY. A
method has been developed to transfer autologous keratinocytes to a wound bed within 5-7
days of receipt of biopsy. With the development by Smith and Nephew of a
polymer that supports cell growth it has been found that actively dividing
(undifferentiated) keratinocytes will invert and grow on a suitable wound bed.
Keratinocytes were grown in flasks in serum containing medium for 48 h, fed with
serum-free medium, and when actively growing were transferred to the polymer. This was
then incubated for 2-3 days to produce a subconfluent cell layer (70-90% confluent) which
was easily inverted on to the wound bed in theatre or at the bedside. When placed on a
suitable wound bed, healing was evident as a translucent film after 5-7 days.
Introduction
Since the development of
keratinocyte cultures in Howard Green's' department in Boston and their use by O'Connor'
to graft severely burned patients, there has been worldwide interest in wound healing by
this method. The Burns Unit at Queen Mary's Hospital, Roehampton has been using
keratinocytes cultured by this method since 1985 .4,5 However, it was found that the
length of time required from biopsy to graft often extended rather than reduced the
healing time.
The use of a polymer film (developed by Smith and Nephew)' compatible with keratinocyte
culture has enabled us to reduce the time required for cell growth, and allowed easier
transfer of cells to the patient than the original method.
Materials and
methods
The original culture
method for keratinocyte grafting has been modified. 15 A thin split-skin biopsy, about 5
sq. cm, was taken under general anaesthetic and transferred to the laboratory for culture.
The sample was washed well in saline and incubated in Dispase (Collaborative Biochemicals
Ltd., Bedford, Ma.) at 37 'C for 30-40 mins. The epidermis was then removed to a standard
trypsin/Versene solution and incubated at 37 'C for 10 min. The cell suspension was
removed and the trypsin neutralized with serum-containing medium before centrifugation.
(The remaining sample was trypsinized a second time and treated like the first.) The
resulting pellets were kept separate (to limit contamination), each being resuspended in
serum-containing growth medium.
The cells were counted in a haemocytometer. Culture flasks were inoculated at 3-5 x 101
cells per ml in growth medium containing serum, and incubated at 36.5 'C for 48 h. (This
medium, based on 3:1 DMEM/1712 with added growth factors and 10% foetal calf serum, was
designed by Green' for keratinocyte growth.) Cultures showing growth at this time were fed
with serum-free medium by replacing half the supernatant with MCDB 153' (Clonetics Corp.,
La Jolla, Ca.)
After a further 2-3 days growth, or 48 h before required for grafting, the keratinocytes
were trypsinized and transferred to polymer discs (35 mm diameter) in 6well plates at 3-5
x 101 cells per disc in 2 ml serum-free medium. The plates were incubated at 36.5 'C in 5%
C02 in a humid incubator.
When the wound had been prepared the cultures were taken to theatre or bedside. The
polymer discs were lifted with forceps and inverted on to the wound. The area was covered
with Jelonet and a dry dressing, and bound firmly with cr&pe bandage.
The sources of media and other solutions were as follows: foetal calf serum and bovine
pituitary extract, First Link UK, West Midlands, UK; Dulbeccos Modified Eagles Medium
(DMEM), Hams F12; Imperial Laboratories, Hampshire, UK; Dispase; Collaborative Biomedical
Products, Bedford Ma., USA; Trypsin/EDTA and Medium Additives, Sigma Biosciences Company,
Dorset, UK.
This method differs from that of Green in several respects:
the epidermis is separated from the
dermis with Dispase before trypsinization
no feeder layer is used, and the
number of cells in the inoculum is increased
after 48 h the cultures are
gradually weaned on to serum-free medium
the cultures are subcultured before
reaching confluence in order to maximise growth
the cultures are easy to transfer. The
polymer discs need only to be lifted from the dish with forceps and inverted on to the
wound
Results
Case 1
A three-year-old victim of a house fire sustained burns to the right side of her head, arm
and leg. Following resuscitation procedures and excision, the leg was grafted with meshed
split-skin from the father. This was left for two weeks before removal of the epidermis,
thorough washing with saline, and coverage of the wound with polymer discs. Autologous
keratinocytes grown on the polymer discs were 60% confluent. At the same time other wounds
were treated with meshed autograft.
At the first dressing change there was evidence of infection in both areas. Topical
antibiotics were applied and washed off with saline after three days. Further cultured
keratinocytes on polymer discs were then applied to the wound.
After a further five days the dressings were removed, showing distinct opaque areas of
epidermis where some of the discs had been placed. Some of the discs had slipped and
further cultures were therefore used to cover the remaining wound on the girl's leg.
The patient was allowed home wearing a Jobst garment and she healed without further
incident (Figs. 1-4).
|
|
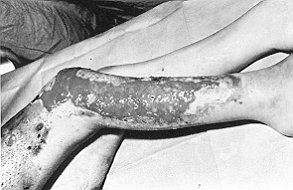
Fig.
I - Case 1. De-epidermidized allografted
wound bed prior to first culture graft. |
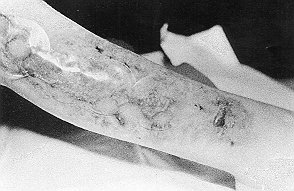
Fig.
2 - Case 1. Wound after second culture graft,
5 days post-grafting, showing opaque healed area
beneath disc. |
|
|
|
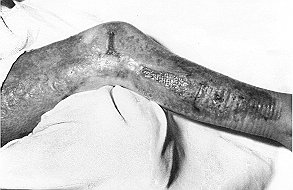
| Fig. 3 - Case 1. Patient ready for release
after 3 weeks |
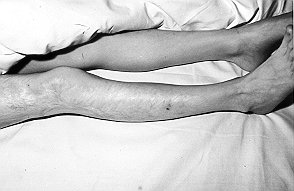
Fig.
4 - Case 1. Appearance of wound after 3 years. |
|
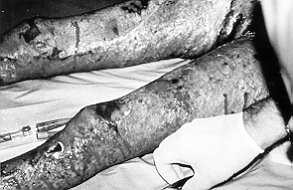
Case 2
This patient was transferred to our Burns Unit three weeks
post-burn. The injury had taken place as she was cooking over an open fire. Ten days
later, after excision, twelve polymer disc autocultures (of 60-90% confluence) were
grafted on to full-thickness wounds on both legs. Seven days later the discs were removed
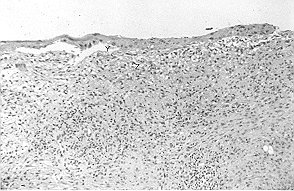
with the dressings. The biopsies taken showed considerable epithelialization (Figs. 5-6).
Unfortunately, the patient died two weeks later.
|
|
| Fig.
5 - Case 2. Right leg 10 days post-grafting showing site of biopsy (x). |
Fig. 6 - Case 2. Biopsy section showing narrow
epidermal layer (y) loosely attached to fascia (z) of wound bed. |
|
Case 3
This patient, aged two years, had 30% skin loss owing to
meningococcal septicaemia. Following excision he was grafted with glycerol-preserved
allograft and five days later this was de-epidermidized and keratinocyte cultures placed
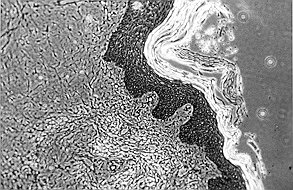
on an allodermal area on each lower leg (Fig. 7).
After a further five days the discs were removed and biopsies taken from each site. The
right leg was infected and both wounds were split-skin grafted. The biopsies showed that
full epidermis had grown on the left leg.
|
Fig.
7 - Case 3. Biopsy 5 days post-grafting on de-epidermidized homograft (stain ME),
showing re-epithelialization. This was not visible macroscopically as differentiation was
incomplete. |
|
Discussion
Novel synthetic polymer films produced by
Smith and Nephew have been shown to support the growth of human keratinocytes. These have
been used to transfer actively growing cells to burn wounds in patients. The film, made in
3.5 cm diameter circles, was impermeable to air and non-adherent to the wound bed.
The theories behind this technique were:
- that cells in a subconfluent culture show more mitotic
activity and a reduced tendency to differentiate than in confluent cultures and cell
sheets
- that actively dividing, undifferentiated cells are able to
invert and transfer from the polymer and grow on a suitable wound bed
By the use of serum-free
low-calcium medium, growth was enhanced and differentiation minimized, so that cells were
able to grow on to the wound bed. This method enabled autologous cells to be grafted
within days of receiving the biopsy instead of the more usual 3-4 weeks' delay of previous
methods. First passage cells were grafted before confluence occurred and they therefore
underwent fewer divisions before application. Differentiation took place on the wound bed,
protected by the polymer, until epidermis was formed and the disc dropped off during a
dressing change.
There were other advantages using this method. Unlike the previous technique, according to
which the cell sheet must be removed from the base of the culture vessel and mounted on a
dressing before transfer to theatre or bedside, transfer of the discs was quick and easy,
requiring little skill to lift (with forceps) from culture dish to wound bed, with no
previous preparation. Problems with transportation over long distances and viability
maintenance are thus minimized using this system, the cultures being kept in growth medium
until grafting has taken place.
From the small number of patients grafted in this trial it has been demonstrated that
cells will migrate from the polymer surface and form areas of healthy epidermis on
suitable wound beds. It is unlikely that this is a 'dressing effect' which stimulates
epidermal migration from adnexal elements or the wound edge, as discrete areas of healthy
epidermis were observed beneath the centre of the polymer discs five days after grafting
over areas of previously allografted, full-thickness wound.
The major causes of graft failure in this study were displacement of the polymer, and
microbial infection entering round the edge of the disc. The condition of the wound bed at
the time of grafting and the presence or absence of dermal elements also affected the
'take' or failure. We hope to combine this polymer/epidermal system with allodermis from
banked skin from the new Stephen Kirby Skin Bank or dermal equivalents currently being
developed.
RESUME.
Les Auteurs ont développé une méthode pour permettre de transférer des kératinocytes
autologues jusqu'au site d'une lésion entre 5-7 jours après la biopsie. Grâce au
développement par Smith et Nephew d'un polymer qui supporte la croissance des cellules,
ils ont trouvé que les kératinocytes (non différenciés) qui se divident activement
peuvent invertir et croître sur une lésion. Les kératinocytes ont été cultivés dans
des éprouvettes dans un milieu qui contenait du sière pendant 48 h, alimentés avec
milieu sans sérum, et transférés, une fois en phase de croissance active, jusqu'au
polymer. Celui-ci a été incubé pendant 2-3 jours pour produire une strate cellulaire
subconfluente (confluence 70-90%) qui était facile à invertir sur le site de la lésion
en salle opératoire ou au chevet du lit. Après la mise en place sur une lésion
appropriée, la guérison était évidente, comme une pellicule translucide, après 5-7
jours.
BIBLIOGRAPHY
- Rheinwald J.G., Green H.: Serial cultivation of strains of
human epidermal keratinocytes: the formation of keratinising colonies from S.single cells.
Cell, 6: 331-44, 1975.
- Gallico G., O'Connor N.E., Compton C.C. et al.: Permanent
coverage of large burn wounds with autologous cultured human epithelium. N. Engl. J. Med.,
331:448-51,1984.
- De Luca M., Albanese E., Bondanza S. et al.: Multicentre
experience in the treatment of burns with autologous and allogenic cultured epithelium,
fresh or preserved in a frozen state. Burns, 15: 303-9, 1989.
- Eldad A., Burt A.M., Clark J.A.: Cultured epithelium as a
skin substitute. Burns, 13: 173-80, 1987.
Burt A.M., McGrouther
D.A.: "The production and use of skin cell cultures in therapeutic situations".
Animal Cell Biotechnology, Spier R.E., Griffiths J.B. (eds), Academic Press, vol. 5:
151-68, 1992.
Barlow Y.M., Burt A.M., Clarke J.A. et al.: The use of a
polymeric film for the culture and transfer of subconfluent autologous keratinocytes to
patients. J. Tiss. Viab., 2: 33-6, 1992.
Cuono C.B., Langdon R., Birchall N.E.: Composite
autologous/allogenie skin replacement: development and clinical application. Plast.
Reconstr. Surg., 80: 626-30, 1987.
Ben-Bassat IL, Eldad A., Chacmat M. et al.: Structural and
functional evaluation of modifications in the composite skin graft: cryopreserved dermis
and cultured keratinocytes. Plast. Reconstr. Surg., 89: 510-20, 1992.
| This paper was received on 20 August
1997. Address correspondence to: Dr
Avril J. Burt
Stephen Kirby Skin Bank, Burns Unit
Queen Mary's University Hospital
Roehampton Lane, London SW15 5PN, Great Britain. |
Acknowledgements. We wish to thank
both Smith and Nephew for their support and provision of the polymer and Dr Y. Barlow
during her employment with them. Thanks also for the help of the nursing staff of the
Burns Unit at Queen Mary's Hospital, Roehampton.
|






