Annals of 1he MBC - vol. 2 - n* 2 -
June 1989
RECENT
FASCIOCUTANEOUS FLAPS FOR REPAIR OF POST-BURN NECK, AXILLARY AND ELBOW CONTRACTURES
Kadry M., Hassan A., Tewfik 0., Ismail M.
Plastic surgery Department, Faculty of Medicine, Cairo
University, Egypt
SUMMARY.
Deep burns are known to result in severe contractures over the neck, axilla and elbow.
After release of the contracture the problem remains to cover the resulting raw area.
Multiple techniques are available to cover such defects, ranging from the use of a
split-thickness skin graft to the most elaborate repair with a free flap. Each of these
techniques has its advantages as well as its hazards and limitations. It used to be the
dream of every plastic surgeon to accomplish this goal by using local tissues which have
the same texture, colour and thickness without the possibility of subsequent contracture.
The fasciocutaneous flaps recently described seem to fulfil this goal. Three flaps
recently described but not widely used are presented. Their anatomical basis, their
clinical applications as well as their advantages and drawbacks are demonstrated for
repair of post-burn axillary, cervical and elbow contractures.
Introduction
Severe contractures of the axilla,
neck and elbow are known to follow deep burns of these regions.
After release of these contractures the resulting raw areas need a suitable covering. The
conventional methods range from the use of split-thickness skin graft, myocutaneous flaps
up to the most complicated repair with a free microvascular flap.
It used to be the dream of every plastic surgeon to use local tissues having the
possibility of recontracture.
Since the evolution of fasciocutaneous flaps, these requirements seem to be fulfilled.
Tolhurst (1983) believed it both fair and logical to retain Ponten's (1981) original
definition of fasciocutaneous flaps (flaps comprising skin, subcutaneous fat and fascia
but no muscles), but classified them into subgroups according to their blood supply.
Type A: dependent on multiple
fasciocutaneous perforators, entering its base and oriented with the long axis of the flap
parallel to the direction of the arterial plexus
Type B: based on a single
fasciocutaneous perforator of a moderate size
Type C has multiple small
perforators along its length which reach it from a deep artery by passing along a fascial
septum between the muscles
Type D: an osteofasciocutaneous
free tissue transfer, an elaboration of Type C.
The aim of this work is to present our
experience with the use of the parascapular flap (Tolhurst and Haeseker, 1982), the
cervicodorsal fasciocutaneous flap (Nakajima and Fujino, 1984), and the reversed ulnar
recurrent fasclocutaneous flap (Maruyama, 1987) for repair of post-burn contractures of
the axilla, neck and elbow respectively.
Material and methods
This series included 39 flaps
distributed as shown in Table 1, as follows:
Posterolateral chest wall fasciocutaneous flap for repair of axilla defects (Tolhurst and
Haesker, 1982) - 14 flaps. Its blood supply is derived from the parascapular artery
(terminal branch of the circumflex scapular artery) which descends along the lateral
border of the scapula passing in the fascial septum between the muscles. It then runs
inferiorly to the skin surface at the level of the deep fascia.
Cervicodorsal fasciocutaneous flap of Nakajima and Fujmo (1984) for repair of cervical
defects - 13 flaps. Its blood supply is derived from the superficial branch of the
transverse cervical artery anastomosing with the posterior branches of the posterior
intercostal vessels.
The reversed uInar recurrent fasciocutaneous flap of Maruyama (1987) for repair of elbow
defects - 12 flaps. Its blood supply is dependent on the u1nar recurrent vessels which run
upwards in the median intermuscular septum.
Axillary Defects Fig. 1 (a, b), Fig. 2 (a, b)
After release of the axillary contracture
a suitable flap with a ratio of 1:3 or more is raised from the posterolateral chest wall
based superiorly and including the fascia over the latissimus dorsi. Upon elevation the
rich vascular network at an epifascial plane is always seen.
 |
 |
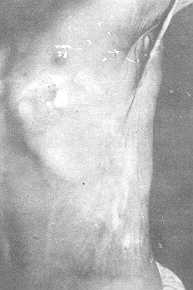
| Fig. 1 (a) -
Severe anterior fold contracture preoperatively. |
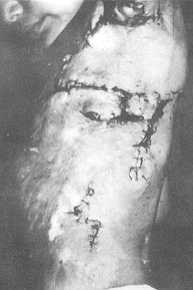
Fig. 1 (b) -
Postoperative view with the flap rotated in place. |
 |
 |
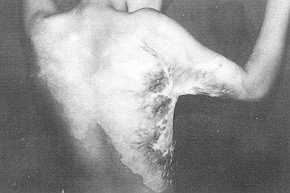
| Fig. 2 (a) - Posterior
axillary full contrpture preoperative. |
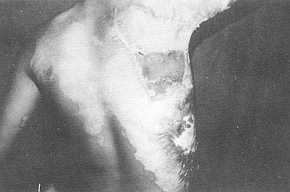
Fig. 2 (b) - Postoperative
view, notice full abduction. |
|
Extensive undermining allows closure of
the donor defect primarily if it was less than 8 cm. An S.T.S.G. was however needed to
close the donor defect site in 6 of the flaps used.
If the flap is to be used bilaterally it should be done in 2 stages and the flaps should
be more anteriorly situated, including the fascia on the serratus anterior as well.
Cervical Defects Fig. 3 (a, b, c), Fig. 4 (a, b)
After release of the neck contracture, the
defect is measured. The patient is put in the lateral decubItus position. The flap is then
designed with its base situated superiorly extending over the upper border of the
trapezius to include the superficial branch of the transverse cervical artery, as an
island flap. The deep fascia over the latissimus dorsi and the trapezius should be
included in the flap. The donor defect can usually be closed by direct sutures, but if
there is undue tension a small piece of S.T.S.G. is used.
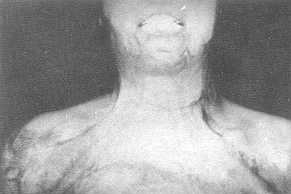 |
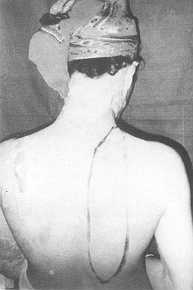 |
| Fig. 3 (a) -
Preoperative view of post-bum neck contracture. |
Fig. 3 (b) - Flap
design |
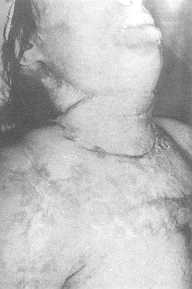 |
| Fig.
3 (c) - Late postoperative view. |
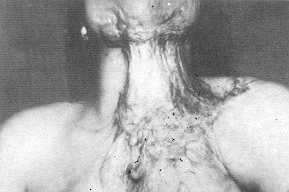 |
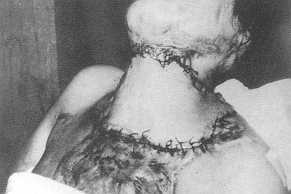 |
| Fig. 4 (a) -
Preoperative view of post-burn elbow contracture. |
Fig. 4 (b) -
Early postoperative view, note neck extension. |
|
Elbow Defects Fig. 5 (a, b, c), Fig. 6 (a, b)
After release of the contracture and
measuring the defect, a flap is designed on the ulnar aspect of the upper arm with its
lateral border between the midanterior and the midposterior lines. The flap is elevated
from proximal to distal as an island including the deep fascia, and dividing the medial
intermuscular septum between the triceps and the brachialis as high as possible, to avoid
injury of the ulnar nerve. Donor site is closed, usually by direct sutures.
Results
Restoration of a full range of abduction was gradual
and progressive over the following 3 months after release of the axillary contractures.
In one case a linear contracture developed at the anterior axillary fold and required a
Z-plasty after 6 months to obtain full abduction.
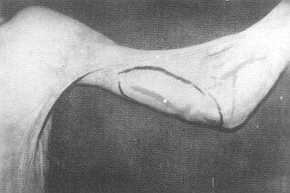 |
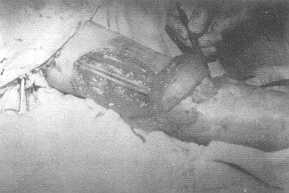 |
| Fig. 5 (a) -
Preoperative view of post-burn elbow contracture. |
Fig. 5 (b) -
Intraoperative view with the flap rotated in place. |
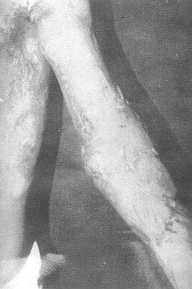 |
| Fig.
5 (c) - Late postoperative view, note full extension of elbow. |
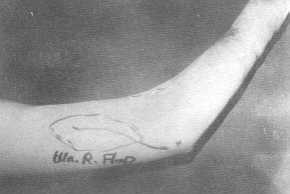 |
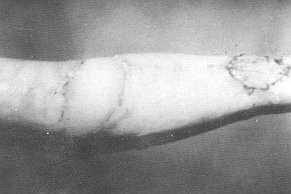 |
| Fig. 6 (a) -
Preoperative post-burn elbow contracture with flap design. |
Fig. 6 (b) -
Late postoperative appearance of fully extended elbow. |
|
The dog ear at the pivot of the posterolateral
fasciocutaneous flap was not complained of, though always present.
The complications of this series are shown in Table 2.
Type of flap |
Number of flaps |
| Posterolateral chest wall for repair of axillary
defects (Tolhurst & Haeseker 1982) |
14 |
| Back island flap for repair of cervical defects
(Nakajima & Fujino 1984) |
13 |
| Reversed ulnar recurrent for repair of elbow
defects (Maruyama 1987) |
12 |
| Total |
39 |
|
Tab. 1 |
|
| |
Neck |
Elbow |
Axilla |
| Total flap loss |
1 |
- |
- |
| Partial flap necrosis |
2 |
2 |
1 |
| Epidermolysis |
2 |
2 |
- |
| Donor site infection |
1 |
1 |
1 |
|
Tab. 2
- COMPLICATIONS |
|
Total Flap Loss
This occurred in the first case of
cervicodorsal flap. The flap, though having good viability on the first day, suftered
cyanosis after 36 hours. It was repositioned and the neck was skin grafted. This loss was
presumably due to kinking of the pedicle (mainly venous) from faulty positioning of the
patient in bed.
Partial Tip Flap Necrosis
This occurred on the distal 2-3 cm of
the flap. In all these cases the flap was originally deeply burned or skin grafted.
Although the deep site of the vascular pedicle allows for the use of flaps in such
circumstances, one should not exceed the length/breadth ratio in such cases. In the
axillary flap which underwent this complication the ratio was 1A.
Proper wound care and minimal debridement achieved healing in 4 patients. In the axillary
case an anterior pulling of the flap and mobilization was required to close the defect.
Epidermolysis
Superficial skin loss with spontaneous epithelization
occurred in 4 cases. These cases had a deep bum at the pedicle site which healed by
cicatrisation.
Donor Site Infections
These all responded to repeated dressings and
appropriate antibiotics.
Discussion and conclusions
The aim of the management of any burn
contracture is to achieve a good functional and, if possible, a good cosmetic result by a
one-stage uncomplicated procedure (Kadry et al., 1985).
After release of axillary, cervical and elbow contractures, though skin graft provides an
easy solution, their take is never complete, and they are liable to recontractures.
Various types of skin flaps usually end with bulky cover that may need defattening and
their donor sites are often involved in the original burn, their use thus being limited.
The use of the latissimus dorsi myocutaneous flap for the covering of such defects became
very popular after its description by Bostwick (1979). However these flaps are too bulky,
unaesthetic and their donor site morbidity is quite significant.
Fasciocutaneous flaps on the other hand are thin, reliable, one-stage procedure, and are
easily harvested without any subsequent functional impairment since the underlying muscles
are left intact (Tolhurst et al., 1983).
After release of an axillary contracture the defect is usually larger than anticipated.
The topography of the axillary pit makes it difficult to properly immobilise any graft
applied. Full arm abduction for 3 months in a cumbersome splint is usually required.
The fasciocutaneous flap seems to obviate much of the trouble. It accomplishes a good
functional and cosmetic result. The donor site, closed by primary suture or skin grafted
in an already burned area, is tolerable. The resulting dog ear is not bothering to the
patient. The good vascular element at the level of the deep fascia allows the use of
scarred skin or skin grafted flap with a 1:3 ratio that could not be used in random flaps.
Exceeding 1:3 ratio however proved very unsafe.
Nakajinia and Fujino (1984) described the vascular basis of the cervicodorsal flap, and
its application in 4 cases. They gave little if any details about the technical details.
One of the common findings in the use of this flap is the development of postoperative
cyanosis which jeopardizes the flap. The following recommendations are given to avoid such
a mishap:
- some of the fibres of the trapezius should be included with
the pedicle
- there is no need to expose the transverse cervical vessels
as this may affect the integrity of the flap
- avoid too tight closure of the skin around the pedicle
- postoperative perfusion of the patient with 3 litres of
lactated Ringer's sol. maintains a good flap circulation
- proper positioning of the patient during the first 3-4
postoperative days is crucial. This is accomplished by putting the patient on his back
with hyperextension of the neck with pillows under the shoulders.
In case of reversed ulnar recurrent
fasciocutaneous flap for the closing of elbow defects there is no need to dissect the
pedicle as this may not only induce its spasm, but also endanger the ulnar nerve itself or
one of its cutaneous branches.
The recurrent ulnar fasciocutaneous flap does not seem to be a useful flap in obese
females. Furthermore, in cases of deep cicatrization over the site of the flap, we
strongly suggest the use of a fascial flap which is split-thickness grafted to avoid
epidermolysis or partial flap necrosis.
In conclusion, fasciocutaneous flaps are reliable and ready solutions to cover defects
after release of post-burn contractures of the axilla, neck and elbow. They should
establish their place as a useful tool for all plastic surgeons.
RÉSUMÉ. Les
brûlures profondes provoquent des contractures sévères du cou, de l'aisselle et du
coude. Après la décharge de la contracture il reste le problème de la couverture de la
zone exposée résultante. On peut utiliser diverses techniques pour remédier à ce
problème, depuis l'emploi d'une greffe cutanée à épaisseur variable jusq'au lambeau
myocutané libre. Chacune de ces techniques présente des avantages comme aussi des
risques et des limitations.
Tous les chirurgiens plastiques voudraient atteindre ce but en utilisant des tissus locaux
qui possèdent la même structure, couleur et épaisseur sans présenter risque de
contracture successive. Ce but, semble-t-il, peut être atteint avec les lambeaux
fasciocutanés. L'Auteur, après avoir présenté trois types de lambeaux récemment
décrits mais pas encore largement utilisés, indique leur base anatomique, leurs
applications cliniques et leurs avantages et inconvénients dans la réparation des
contractures à la suite des brûlures du cou, de l'aisselle et du coude.
BIBLIOGRAPHY
- Bostwick J., Nahai F., Wallace J.G., Vasconez L.O.:
Sixty Latissimus Dorsi Flaps. Plast. Reconstr. Surg., 63: 31-41, 1979.
- Kadry M., Gat6 A., Bernelmans D.: The Cicatricial
Sequelae of Extensive Trunkal Burns (Topographical Classification & Proposal of a Plan
of Treatment). Egypt. J. Plast. Reconstr. Surg., 9: No 2, 1985.
- Maruyama Y.; Onishim K., Wahira Y.: The Ulnar
Recurrent Fasciocutaneous Island Flap: Reversed Medial Arm Flap. Plast. Reconstr. Surg.,
79: 381-384, 1987.
- Nakajima H., Fujino T.: Island Fasciocutaneous Flaps
of Dorsal Trunk and their Application to Myocutaneous Flap. Keio J. Med., 33: 59-82, 1984.
- Ponten B.: The Fasciocutaneous Flap: Its Use in Soft
Tissue Defects of the Lower Leg. Brit. J. Plastic Surg., 34: 215-220, 1981.
- Tolhurst D.E., Haeseker B., Zeeman R. J. : The
Development of Fasciocutaneous Flap and its Clinical Application. Plast. Reconstr. Surg.,
71: 597-605, 1983.
- Tolhurst D.E., Haeseker B.: Fasciocutaneous Flaps in
the Axillary Region. Brit. J. Plast. Surg., 35: 430-435, 1982.
|













