Annals of the MBC - vol. 3 - n' 3 -
September 1990
DUODERME IN THE
TREATMENT OF DONOR SITES: A REPORT
Hermans M.H.E.
Convatec Northern Europe, The Netherlands
SUMMARY. In a prospective study in
two burn centres and one rehabilitation centre DuoDERM E, a new hydrocolloid dressing, was
tested as a dressing for the treatment of donor sites. The result of these tests prove
that DuoDERM E provides fast reepithelialization of the wounds, good cosmetic results and
patient comfort.
Introduction
Although donor sites usually heal
without many problems, they are very painful to the patient. Pain relief can be obtained
using occlusive dressings. With one of these dressings, DuoDERM, fast healing, good
quality of the healed wounds and pain reduction is reported in many articles (2, 4, 8,
11).
The clinical use of DuoDERM in highly exudative wounds such as donor sites however
sometimes causes practical problems, as leakage of the gel which is produced by the
dressing makes it necessary to change the dressing quite often.
The gel produced by DuoDERM E has different physical properties. The aim of the two
clinical studies described in this report was to assess these properties and their
influence on the quality of wound healing, the healing time, the number of dressings
necessary before healing is complete and patient comfort.
Materials
DuoDERM E is a second generation
hydrocolloid dressing in which a stronger binding of the gel to the dressing matrix is
obtained by incorporating the same hydrocolloids as in DuoDERM in a difterent polymer.
From a theoretical point of view, less leakage will occur and less frequent dressing
changes will be necessary. The outer layer of DuoDERM E is made of a polyurethane foam
which is impermeable for oxygen, vapour, gases and micro-organisms. The dressing is
adherent to normal skin. When applied over healthy skin, the wound side of the dressing
adheres very well: in general no additional bandages are needed to keep DuoDERM E in
place.
Methods
Patients in the Bum Centre of the H6pital de la
Conception in Marseilles (France), the Rehabilitation Centre H6pital Leon 136rard in
Hy&res (France) and the Burn Centre of the Stuivenberg Hospital in Antwerp (Belgium)
participated in this study.
Donor sites were covered with DuoDERM E immediately after harvesting the skin, overlapping
the wound on each side with at least 2.5 em, so that there would be enough healthy skin
available for adherence.
DuoDERM E is not transparent. For that reason it is not possible to determine exactly the
rate of reepithelialization without removing the dressing. Two different regimes were used
for dressing changes. In the French centres dressings were changed at a 5 days' interval.
Thus, the number of dressings until reepithelialization could be calculated. In the
Antwerp Burns Centre a new dressing was applied every day after post-operative day 5 to
enable the investigator to assess the moment of complete reepithelialization as accurately
as possible. In both studies pre-operative disinfection was performed with Betadine. In
both centres donor sites were made with an electrical or air-driven dermatome, set to
0.012 inch.
Data to be evaluated during the trial were age and sex of the patient, localization of the
donor site, TI1SA (only for the patients in the burn centres), reepithelialization time
(Belgian centre), the amount of dressings necessary (French centres) and patient comfort.
In all three centres the donor sites were evaluated after three months.
Results
Burns Centre of Marseilles and
Rehabilitation Centre of Hyres
20 patients (10 male, 10 female), of
whom 13 were from the Burns Centre in Marseilles and 7 from the Rehabilitation Centre in
Hy6res, with 28 donor sites, participated in the trial. One patient was operated upon on
two different dates and in 7 patients two donor sites were created during the same
operation.
The average age of the patients was 41.1 years (min. 15, max. 80). The average TBSA on
admission in the patients from the Burn Centre was 12.8% (min. 4%, max. 20%).
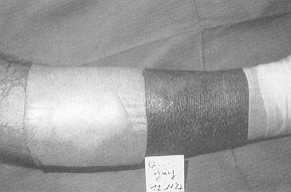 |
 |
| Fig. 1
The gel of DuoDERM E stays located around the wound, without leakage. |
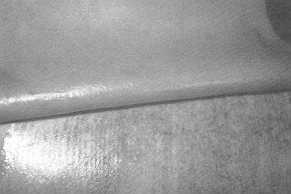
Fig. 2 Post-operative
day 10, second dressing, in situ for 5 days. Although the wound environment is
moist, there is only a very thin layer of gel. |
|
The localizations of the donor sites are
stated in Tab. 1. The average size of the donor sites was 231 sq. cm (min. 24, max. 500).
The reepithelialization time was less than 10 days in 17 donor sites (63%), less than 15
days in 10 donor sites (33%) and longer than 15 days (17 days) in I donor site (4%). On
average 2.3 sheets of DuoDERM E were necessary to achieve complete reepithelialization
(min. 2, max. 5).
Pain during dressing changes and overall comfort of the dressing are described in Tab. 2.
Burns Centre of Antwerp
In this centre 21 patients (15 male, 6
female) with 22 donor sites participated in the trial. The average age of this group of
patients was 34.4 years (min. 2, max. 79). The TBSA of the patients was 8.4% on average
(min. 0.5, max. 34).
The average size of the donor sites was 115 sq. cm (min. 40, max. 322). The localizations
of the donor sites are described in Tab. 1.
To be able to assess the healing time in this group, after post-operative day 5 the
DuoDERM E was changed every day. The average reepithelialization time was 8.1 days (min.
5, max. 15).
Pain during dressing changes and overall comfort of the dressing are described in Tab. 2.
All centres
Three months after reepithelialization
only one (1.6%) out of all 59 donor sites in both groups developed an area of hypertrophy,
less than 0.5 cm in diameter.
|
Burns Centre
Marseilles
Rehabilitation Centre
Hyères |
Burns Centre
Antwerp |
Upper arm:
Forearm:
Calf..
Flank:
Thigh:
Buttock |
4
2
4
2
16
-
28 |
-
-
-
-
19
3
22 |
|
Tab. I
NUMBER OF LOCALIZATIONS OF THE DONOR SITES |
|
| PAIN DURING DRESSING CHANGES |
| |
|
Not
Painful |
Moderately
Painful |
Very
Painful |
| Marseilles/Hyères |
(N=44) |
28(64%) |
16(36%) |
--- |
| Antwerp |
(N= 100) |
94(94%) |
5(5%) |
1(1%) |
| OVERALL DRESSING COMFORT |
| |
|
Very
Comfortable |
Moderately
Comfortable |
No
Opinion |
| Marseilles/Hyères |
(N=21) |
18(86%) |
3(14%) |
--- |
| Antwerp |
(N= 21) |
9(42%) |
9(42%) |
3(16%) |
|
Tab. 2
PAIN DURING DRESSING CHARGES AND OVERALL COMFORT |
|
Clinical signs of infection did not occur
in any of the donor sites. In none of the patients did treatment of the donor sites with
DuoDERM E have to be stopped prior to complete reepithelialization.
Discussion
Although donor sites in general heal
with fewer problems than burns, they should be regarded as wounds that deserve optimal
treatment. It has been shown that the occluding of wounds leads to better healing than
exposure to air (14). Therefore, occlusive dressings should also be used for the treatment
of donor sites, instead of the still often used materials like tulle gauze which, upon
removal, cause destruction of the woundbed and pain to the patient (13).
The study in Marseilles and Hy~res was aimed at evaluating the patient comfort aspects of
the dressing and the number,of dressings necessary to obtain complete reepithelialisation.
On average 2.3 sheets of DuoDERM E were necessary. Dressing changes in general were not
very painful and treatment of donor sites with DuoDERM E as a whole was judged to be
comfortable to moderately comfortable (Tab. 2).
In the Antwerp part of this study, the aim was to assess reepithelialization time with
DuoDERM E, although other properties of the dressing were assessed too. An average
reepithelialization time of 8.1 days, as found in this study, is comparable to the results
with the regular DuoDERM found in other studies (3, 8, 11). In this study dressing changes
were in general considered not painful (94%) and the overall patient comfort was high to
moderate, although 3 patients gave no opinion (Tab. 2). In fact, these results may be
negatively influenced by the fact that, for reasons of judging the wound healing process,
the dressings were changed every day after post-operative day 5, which obviously
interferes with the normal activities~ and overall comfort of the patients.
Treatment with DuoDERM E did not have to be stopped before reepithelialization was
complete in any of the donor sites. One donor site (1.6%) out of all 59 showed a very
small area of hypertrophy after three months.
Conclusion
The studies described in this article
have shown that the good results in the healing of donor sites that can be obtained with
DuoDERM can be equalled by the results with DuoDERM E, as this new material leads to fast
and cosmetically good wound healing. The studies also have shown that with DuoDERm E these
results can be obtained with a minimum of dressings and high patient comfort.
The Author wishes to thank Dr C. Echinard (Marseilles and Hy~res) and Dr R. Peeters
(Antwerp) for their clinical work in these studies. Both studies were sponsored by
Convatec.
Other marketed names for DuoDERM E are DuoDERM Control Gel Formula, DuoDERM CG17,
Granuflex E and Varihesive E.
RÉSUMÉ. Dans une étude
prospective menée chez deux centres de brûlés et un centre de réhabilitation, le
DuoDERM E, un nouveau pansement hydrocolloïde, a été testé dans le traitement des
zones donatrices de la peau. Les épreuves effectuées indiquent que le DuoDERM E offre
une rapide réépithélialisation des plaies et une bonne qualité des résultats
cosmétiques, avec un bon confort pour les patients.
BIBLIOGRAPHY
- Alvarez O.M., Mertz P.M., Hefton J.M., Eaglestein
W.H.: The effect of occlusive dressings on collagen synthesis and reepithelialisation in
superficial wounds. J. Surg. Res, 35: 142-148, 1983.
- Alvarez O.M., Gefton J.M., Eaglestein W.H.: Healing
wounds, occlusion or exposure. Infection in Surgery: 173-178, 1984.
- Biltz H., Kiessling R., Kreysel H.W.: Comparison of
hydrocolloid dressing and saline gauze in the treatment of skin graft donor sites. In:
Ryan T.J., Ed., "An environment for healing: the role of occlusion", Royal
Society of Medicine, 136: 125-128, London, 1985.
- Donati L., Vigano M.: Use of the hydrocolloidal
dressing DuoDERM for skin donor sites for bums. Int. J. Tissue React., 10: 267-272, 1988.
- Doherty C., Geraldine L., Noble S.: Granuflex
hydrocolloid as a donor site dressing. Care of the critically ill, 5: 11-12, 1986.
- Eisenberg M.: The effect of occlusive dressings on
reepithelialisation of wounds in children with epidermolysis bullosa. J. Pediatr. Surg.,
21: 829-894, 1986.
- Hermans M.H.E., Hermans R.P.: DuoERM, an alternative
dressing for smaller bums. Bums. 12: 214-219, 1986.
- M , adden M.R., Finklestein J.L., Hefton J.M, Yurt
R.: Optimal healing of donor sites with hydrocolloid dressings. In: Ryan T.J. Md.,
"An environment for healing: the role of occlusion", Royal Society of Medicine,
136: 133-137, London, 1985.
- May S.R.: Physiology, immunology and clinical
efficacy of an adherent polyurethane dressing: Opsite. In: Wise L.W. Ed., "Bum Wound
Coverings", Vol. 11., CRC Press Inc, 53-78, New York, 1984.
- McHugh T.P. et al.: Therapeutic efficacy of Niobrane
in partial and full thickness thermal injury. Surgery, 100: 661-664, 1986.
- Perrot J., Carsin H., Gibaud J. Utilisation du
pansement DuoDERM dans la cicatrisation des prises de greffes chez le brdl~. Arm. Chir.
Plast. Esthet., 31, 3: 279-282, 1986.
- Waffle C, Simon R.R., Joslin C.: Moisture vapour
permeable film as an outpatient burn dressing. Burns, 14: 14-70, 1988.
- Winter C.D.: Healing of skin wounds and the
influence of dressings in the repair proces. In : Harkiss K.J. Ed., "Surgical
dressings and wound healing", Bradford University Press, 46-77, Bradford, 1970.
- Winter G.D.: Effect of air drying and dressings on
the surface of a wound. Nature, 197: 91-93, 1963.
|

