| Ann. Medit. Burns Club - vol. VII - n, 2 - June 1994
BACTERIOLOGICAL MONITORING IN A BURNS CENTRE P. Cucchiara, M. Sucameli, M. Masellis, M.V. Torregrossa*, L. Valentino*, A. Carmeni** Divisione di Chirurgia Plastica e Terapia
delle Ustioni, Ospeclale Civico, Palermo, Italy SUMMARY. On the basis of the observation of multiresistant strains of Staphylococcus aureus in patients admitted to the Palermo Bums Centre, it was decided to consider the role played by the environment as a "reservoir" of pathogens. A microbiological monitoring programme was undertaken regarding all elements contributing to the circulation of germs around the patient such as the medical, paramedical and auxiliary staff and everything that can serve as a vehicle of infection (air, water, surgical in , struments, disinfectants and the rooms in the Centre). The microbial monitoring protocol that was thus prepared for the personnel and the environment was judged to be effective Introduction In a burns centre intensive care unit,
infectious complications are a possibility that is feared but also sometimes preventable,
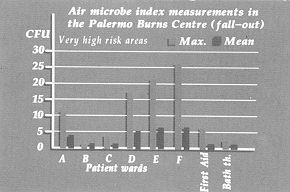
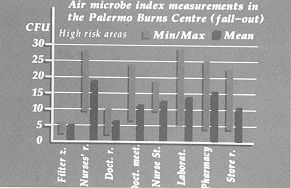
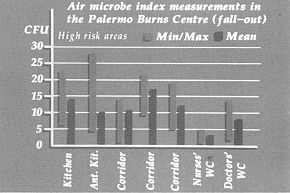
and a considerable cause of morbidity and death. Materials and methods Monitoring was performed in the wards, with simple isolation, and in all rooms used by health personnel working in the Department (Fig. 1, Plan of the Centre). The microbial contamination of the air was assessed by determining the Air Microbe Index (AMI, Pitruzza, 1984; reference values given in Table 1) (1, 2, 3, 4). A total of 396 AMI measurements were performed in the Intensive Care Unit rooms both after environmental
* This level must not be exceeded in any part of the hospital. Three 10-cm diameter petri dishes
containing nutrient agar, blood agar and Sabouraud's agar were exposed in each room for
one hour, one metre above the ground and one metre from every obstacle (Fischer's 1 - 1 -
1 scheme). The dishes were incubated at 37 C and at 22 C and read after 24-48 h for
bacteria and after 72h for fungi. For each room the mean dish values are given as colony
forming units (CFU) (5, 6, 7).
For the health personnel, monitoring was carTied out by a culture test of the material from nasal and pharyngeal swabs and by examination of surgical gloves before and after contact with the patient and the environment surTounding the patient. This test was performed on all medical, nursing and auxiliary staff (10, 11). The monitoring of the patients was performed on hospitalized subjects with deep burns in 30-70% body surface area by means of surface swabs from burned and immediately adjacent areas, biopsies and haemocultures that were seeded on culture media: ordinary broth, simple agar, blood agar, bromothymol blue lactosate agar and Pseudomonas agar. The strains were identified by morphological staining and biochemical techniques, and all the isolated strains were tested to assess their sensitivity to various antibiotics (12, 13, 14, 15, 16, 17, 18, 19). Results Monitoring of the environment Table III - IV - V - W indicates the mean values of all the measurements made in the various rooms in the Burns centre, and the maximum and minimum values obtained: the recommended limit was only occasionally exceeded in very high risk areas (first-aid room, wards, bath therapy rooms) and in high risk areas (laboratory, kitchen, corridors, store-rooms and pharmacy, staff-rooms, etc.). In this last category the highest values were found in the laboratory, the kitchen and the nurses' room. The mean values never exceeded the value in the high risk areas.
Table VII lists the fungi observed. Some fragments of the climatization equipment filters were also examined: Penicillium and Aspergillus niger were observed as well as Clostridia and Staphylococcus lentus, Saprophyticus, Warneri) (5, 20, 2 1).
Strains of multiresistant S. aureus were isolated in the first investigation: they were found once only in a patients' room, while they were always present in the doctors' meeting room, the nurse-station, the laboratory, the corridor and even the filter zone. Many of these strains are methicillin-resistant and in the last test one of these, with the same biochemical characteristics and sensitivity to antibiotics, was isolated from the blood of a young patient in a state of sepsis. The same strain was isolated a month later in the nurse-station (12, 14, 15, 16, 17, 18, 19, 22) (Fig. 2). Monitoring ofpatients Tests on ten patients showed the presence of strains with the same resistance characteristics (although they were sensitive to vancomycin) as those isolated in the air. Considerations and conclusions The investigation gave rise to a number of considerations.Most of the cases of sepsis occurring in the Burns Centre are due to the circulation of strains of methicillinresistant Staph. aureus on patients subjected to continuous infusion therapy using intravenous plastic catheters. The observation of sedimented germs (fall-out) in the culture dishes of a strain which has good resistance to the external environment (air) and is always present in the different periods of the survey suggested to us that we should perform a "mapping" in time of the air-personpatient system involved in the recirculation of the microorganism, without underestimating the importance of "alerting" germs even in the absence of a high environmental microbic charge. There is certainly a circulation of selected, multiresistant strains capable of causing pathologies in patients who have been undergoing invasive manoeuvres or in severely burned patients in whom the damaged skin barrier allows the entry of any micro-organism into the circulation. With regard to the patients it should be stressed that isolation showed its greatest efficacy in reducing the onset of exogenous infections when there was strict observation of all the preventive measures that interrupt contagion from the environment to the patient, with sterilization of all objects introduced into the rooms and with the use of protective clothing (caps, mask, coats, gloves, overshoes) by staff and visitors. RESUME. Les auteurs, après avoir observé la présence de souches multirésistantes de Staphylococcus aureus chez certains patients hospitalisés dans le Centre de Brûlés de Palerme, ont considéré le rôle joué par le milieu comme "réservoir" de pathogènes. Ils ont initié un programme de surveillance microbiologique sur tous les éléments qui contribuent à la circulation des germes autour du patient, comme les médecins, les infirmiers et le peisonnel auxiliaire, et sur tout ce qui peut agir comme véhicule d'infection comme l'air, l'eau, les instruments, les désinfectants et les services qui font part du Centre. Ils ont préparé un protocole de surveillance microbienne du personnel et du milieu qu'ils ont trouvé efficace. BIBLIOGRAPHY
|
||||||||||||||||||||||||||||||||||||||