| Ann. Medit. Burns Club - vol. VII - n. 3 - September 1994
ALTERED NEUTROPHIL FUNCTION IN PATIENTS WITH MAJOR BURN
TRAUMA
Pralat U.*, Lips U.*, Pallua N.**, Stangel W.'
Department Anaesthesiology* Clinic of Plastic and
Reconstructive Surgery**
Blood ban k- I mm un e haematology***
Hannover Medical School, Hannover, Germany
SUMMARY. After severe burn
injury, large amounts of tissue debris, endotoxins and micro-organisms have to be
eliminated. Growing exhaustion of the non-specific immune system leads to the secondary
immunodeficiency syndrome with polymorphonuclear leucocytes (PMN) profoundly suppressed.
Twenty-seven burn patients were studied with a total of 221 measurements. The results
showed that chemotaxis in the surviving patients was significantly higher than in the
non-surviving group. Superoxideanion and hydrogen peroxide play a suspected role in the
pathogenesis of the adult respiratory distress syndrome (ARDS). However, hydrogen peroxide
production after stimulation with formyl-methionyl-leucyl-phenylalanin (FMLP) was nearly
three times higher in non-surviving patients than in survivors. Patients with a regular
clinical course show lower superoxideanion generation than non-survivors after stimulation
with FMLP. These results indicate that non-surviving patients displayed reduced
chernotaxis, while the generation of possibly damaging oxygen radicals was significantly
enhanced in comparison with patients with a regular clinical course.
Introduction
Acute thermal injury produces a
variety of adverse effects in humans. Paramount among these effects is a dramatic
depression in host resistance such that the burn patient is susceptible to infection by a
number of opportunistic pathogens. Intensive studies over the past several years have
shown that this increased incidence of infection is due to defects in non-specific defence
functions, such as neutrophil and macrophage phagoeytosis (20, 22) and bacterial activity
(2), as well as to an inhibition of specific immune functions, such as cell-mediated
immunity (17, 20, 3 1) and antibody formation (2, 13, 17). Available evidence shows that
although specific immune functions play an equally important role in the patient's defence
network, the nonspecific neutrophil and macrophage activities are immensely important in
controlling infection in the burn patient.
Sepsis and the adult respiratory distress syndrome (ARDS) continue to be major
life-threatening complicalions in patients with extensive thermal injury (1, 14). The
decrease in host resistance to infection is obviously related to a depression of both
humoral and cellular components of the host defence system. Although most parts of the
immune system that have been studied after thermal injury show impairment, cell-mediated
immunity turned out to he most consistently and profoundly suppressed (4, 8).
Polymorphonuelear leucocytes (PMN) form a major part of the non-specific first line of
defence. PMN are a potential source of oxygen free radicals, which are produced in
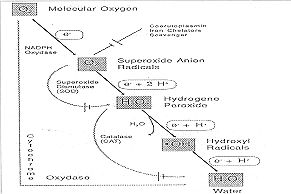
connection with the respiratory burst of these pha~ gocytic cells (Fig. 1). Intracellular
killing by PMN requires the uptake of oxygen by the phagocytic cell. The cytoplasm of the
leucocytes is protected from oxygen free radical damage by the presence of the so-called
radical scavengers, superoxide dismutase and catalase. Through a series of enzyme
reactions including NADPH oxidase, superoxide dismutase and catalase, the oxygen is
progressively reduced to superoxide, hydrogen peroxide, hydroxyl ions and singlet oxygen.
If, however, oxygen free radicals leak into the interstitial space where the concentration
of scavengers is very low, damage to cell membranes is very likely to occur. Chemotaxis is
presumed to be an important function of PMN in protection against invasive agents. Even
leucocytes fully capable of ingesting and killing micro-organisms, tissue debris and
endotoxins can be incapacitated if unable to sense and be directed toward the antigen. In
order to characterize secondary immunodeficiency following severe thermal injury, we
investigated PMN ability as regards chemotaxis and the generation of supetoxideanion and
hydrogen peroxide iii surviving and nonsurviving patients.
|
Fig. 1 - The
normal and monovalent pathway for reduction for molecular oxygen and the enzymatic
defence mechanisms detoxifying the radicat intermediates. |
|
Materials and methods
Patients: in all, 27 patients
suffering from second- and third-degree burns in more than 35% BSA (maximum 92%) were
studied. There were 19 male and 8 female patients ranging in age from 19 to 68 years (mean
age 39.7 ± 9.7 yr). A total of 149 measurements were analysed from the group of 16
surviving patients, and 72 samples were taken from the 11 non-surviving patients, who
eventually died because of sepsis and ARDS. In this group there was an APACHE 111 (11)
score of 76.7 ± 22.8. Nine patients also suffered from inhalation trauma. The surviving
patients had an APACHE Ill (11) score of 40.2 ± 12.8. No patient developed sepsis and
only three were observed to have inhalation trauma.
Informed consent was obtained from all patients or their relatives.
Scrum samples
Samples were taken daily on 11
consecutive days. We investigated PMN chemotaxis against
formyl-methionylleucyl-phenylalanin (FMLP) and yeast-activated serum (YAS) according to
Chenowth (3) in "under agarose technique". The ability of PMN ability to
generate H202 and 02 - was tested after stimulation with opsonized zymosan, FMLP and
zymosan-activated serum (16, 27). Evaluation Of 02- generation was performed by observing
cytochrome C reduction. Assessment of H202 production was carried out kinetically, using
horseradish peroxidase mediated reduction of scopoletin. Statistical analyses were
performed utilizing Student's t-test after establishing medians in each series.
Results
A total of 27 patients were studied,
involving a total of 221 measurements. Matching normal volunteers were used as controls.
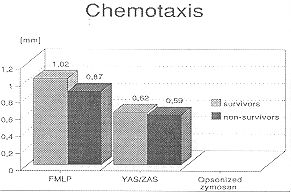
Results are shown in Figs. 2-4. Chemotaxis in response to FMLP in cells from
surviving patients was 1.02 ± 0.27 mm and significantly higher than in PMN from the
non-surviving patients, where it reached 0.67 ± 0.22 mm (Fig.2). There was no significant
difference between the two groups using YAS (surviving group cells, 0.62 ± 0.20 min;
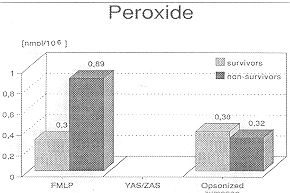
non-surviving group cells, 0.59 + 0.24 min, p<0.001). Fig. 3 shows 11,0,
production after stimulation with FMLP and opsonized zymosan. In PMN from the
non-surviving patients there was a nearly threefold higher H202 generation (0.89 ± 0.19
nmol/106) than in PMN from surviving patients (0.3 ± 0.09 nmol/106) wh 1 ile using FMLR
There was no difference between the two groups after stimulation with opsonized zymosan.
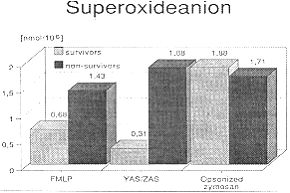
Fig. 4 indicates a significant difference between superoxideanion generation
between the two groups of patients. Surviving patients showed lower 02- production (0.68
± 0.19 mnol/106) than non-surviving septic patients (1.43 ± 0.43 nmol/106) while
stimulated with FMLP. After YAS/ZAS stimulation we find in survivors less 0, - generation
(0.31 ± 0.07 nmol/106) than in non-survivors (1.88 ± 0.24 nmol/106).
Discussion
Severe burns are always followed by
infections which may lead to sepsis, ARDS and multi-organ failure. The complications are
considered to be closely related to the post-burn immunodeficiency syndrome (14).
Increased suspectibility to infections in burn patients occurs in severe conditions,
namely the open contaminated wound, increased metabolic requirements, decreased
nutritional intake, loss of plasma protein and suppression of the patients' defence
mechanisms (8, 15). However, defects do not occur in isolation. As a result it is often
difficult to determine whether a particular abnormality is the primary event, an
impairment which is the consequence of another host defence abnormality, or the result of
infections, metabolic derangements or other secondary effects of the burn trauma.
Based on the results of immunological research, activated leucocytes are now regarded not
only as functioning in specific and nonspecific immune defences, but also as having a
great potential for causing deleterious effects on the host organs.
Neutrophils (24) and macrophages are thought to play especially important key roles in the
pathogenesis of acute lung failure after sepsis and trauma. By an excessive liberation of
lysosomal enzymes, vasoactive eicosanoids and toxic oxygen derived radicals they can cause
increases in permeability, changes in vascular tonus, and damage to the endothelium. of
the lung vessels (6, 9, 10, 19, 29, 30).
|
Fig. 2 - PMN
chemotaxis after stimulation with FNILP and YAS. |
|
As shown by many
investigations, the sequestration of PMN in the lung is a crucial prerequisite for the
full expression of the permeability lesion in ARDS. During lung failure one can regularly
find an excess of adherent PMN in the pulmonary microvasculature, lung lymph and
bronchoalveolar lavage. It has been demonstrated that, in the bronchoalveolar lavage of
patients with ARDS, neutrophils constitute nearly 70% of the recovered cells in comparison
to 14% in normal volunteers and ventilated control patients (28). The percentage of
neutrophils also correlated with A,D02 and lung protein permeability.
The role of polymorphonuclear leucocytes in host resistance to infection is receiving much
attention at the present time. Immediately after antigen stimulation, i.e. by bacteria and
injured tissue, PMN adhere to blood vessel walls and then emigrate into the tissues. This
movement, chemotaxis, is unidirectional, with no return of the cell to the circulation,
and it is facilitated to a large degree by complement. Following phagocytosis, the
intracellular destruction of phagocytized bacteria by PMN is realized by a
"respiratory burst" of cellular activity. However, the ability of PMN to
generate superoxideanion and hydrogen peroxide is extremely interesting as regards
respiratory failure (14).
Our findings in severely burned patients indicate the quantity and quality of parts of
granulocyte impairments. In severely compromised burn patients with fatal outcome, FMLP
mediated superoxideanion production was augmented and correlated with the burned body
surface area and septicaernia (Fig. 4). A similar increase in H202 generation ican
also be observed in this group (Fig. 3).
The reason for this may be a possible defect in detoxification of oxygen radicals with
its suspected role in the pathogenesis of ARDS. After activation of neutrophils, the cells
undergo a "respiratory burst" with an associated increase in oxygen consumption.
In conjuction with this, neutrophils have shown to secrete both superoxideanion and
hydrogen peroxide. Obviously, patients suffering from sepsis following burns show
alteration in oxygen metabolisms at an early stage. Granulocyte stimulation with different
agents proved that the amount of superoxideanion production depends on the kind of
stimulus (5). Tschaikowsky and Ruegheimer (25) have shown that in septic patients there is
an increase in FMLP receptors. It is thought that the integration of pre-formed receptors
following activation of PMN by intrinsic or extrinsic factors or micro-organisms might be
responsible.
Our patients with fatal outcome display compromised chemotaxis to FMLP, while the
generation of possible damaging oxygen radicals is significantly enhanced in comparison to
patients with a regular clinical course. Defects in neutrophil bactericidal capacity were
reported by Alexander in 1971 (2) to be evident in burn patients during periods of
septicaemia. We found that reduced chemotaxis confirms the study of Warden et al. (26),
who described leucocyte chernotaxis as being inversely correlaled with burn size during
the first 72 hours post-burn. After 72 hours, leucocyte chernotaxis directly correlates
with clinical status and is highly predictive for mortality (26). In view of the small
number of patients that we studied, we are not able to confinn this, but since mortality
in our patients was largely due to infection, these findings suggest that suppression of
leukocyte chernotaxis may explain the susceptibility to opportunistic infection in
thermally injured patients. Grogan (7) monitored neutrophil ingestion and bactericidal
capacity in 35 burn patients, using Pseudomonas aeruginosa as the test micro-organism, and
found that defects were common in the phagocytes of patients with burns in more than 30%
BSA. However, for the first five days, there was no acute alteration. After this initial
period, phagocytosis and killing acti-vity were significantly reduced. Nevertheless, 77%
of the septic patients exhibited a reduced neutrophil bactericidal capacity (7). In
general, killing effects were detected more often than an effect in the ingestive phase of
phagocytosis, although the latter was discovered in a statistically significant number of
tests (7).
|
Fig. 3 - Hydrogen
peroxide production after stimulation with FNILP and opsonized zymosan. |
|
In agreement with Alexander
et al. (1) and Solomkin et al. (23) our study shows a decrease in neutrophil
bactericidal capacity which was higher in non-surviving patients. These patients died
because of respiratory failure and sepsis following heavy wound colonization.
Other investigators have reported normal neutrophil function in patients who were severely
ill with septicaemia. Messner et al. (12) and Solberg (22) studied non-burned
patients with infections whose transient neutrophil bactericidal defects returned to
normal when the patients had recovered from the infection.
However, the pathophysiology of decreased chemotaxis and increased metabolic response
remains to be elucidated and a casual relationship between our findings and subsequent
infection can only be theorized. We believe that monitoring chernotaxis and superoxide and
hydrogen peroxide provides an indirect measure of part of the basic membrane and
cytoskeletal functions upon which stimulated migratory functions are based. Speculating
further, one miight expect that therapy to control stimulated secretory responses of the
circulating PMN could effectively limit the generalized cellular dysfunction that
accompanies thermal injury with its excessive complement activation and release of
cytokines.
|
Fig. 4 - Superoxideanion
stimulation with FNILP, ZAS and opsonized zymosan. |
|
RESUME. A la
suite des brûlures sévères, il faut éliminer de grandes quantités de débris
tissulaires, d'endotoxines et de micro-organismes. L'épuisement croissant du système
immunitaire porte au syndrome d'immuno-déficience secondaire, avec les leucocytes
polymorphônucléaires profondément supprimés. Les auteurs ont étudié 27 patients en
réalisant en tout 221 tests. Les résultats montrent que le chimiotactisme était
significativement plus haut chez les patients survivants que chez les patients décédés.
Il est possible que le superoxideanion et l'eau oxygénée jouent un certain rôle dans la
pathogénie du syndrome de détresse respiratoire adulte. Cependant, la production d'eau
oxygénée après la stimulation avec la formyl-méthionyl-leucyl-phénylalanine (FMLP)
chez les patient décédés était presque trois fois plus que chez les patients
survivants. Après la stimulation avec la FMLP les patients avec un cours clinique
régulier montraient une génération de superoxideanion plus basse que les patients
décédés. Les résultats indiquent que les patients décédés avaient un chimiotactisme
réduit, tandis que la génération des radicaux d'oxygène potentiellement nuisibles
était significativement augmentée par rapport aux patients avec un cours clinique
régulier.
BIBLIOGRAPHY
- Alexander JW., Diogigm R., Meakins J.L.: Periodic
variation in the antibacterial function of human neutrophils and its relationship to
sepsis. Ann Surg, 173: 206-13, 1971.
- Alexander JW., Stinnett J.D., Ogle C.K.: Alterations
in neutrophil function. In: Ninnemann J.L. (Ed.): "The immune consequences of thermal
injury". Williams and Wilkins, Baltimore, 1981.
- Chenowth D.E., Roue J.G., Hugh T.: A modified method
for che motaxis under agarose. J. Immunol. Methods, 25: 337-53, 1979.
- Deitch E.A., Landry K.N., McDonald J.C.: Postbum
impaired cellmediated immunity may not be due to lazy lymphocytes but to overwork. Ann.
Surg., 201: 793-802, 1985.
- Fantone J.C., Ward P.A.: Role of oxygen-derived free
radicals and metabolites in leukocyte-dependent inflammatory reaction. Am. J. Path., 107:
397-418, 1982.
- Flick M.R., Hoeffel J., Staub N.C.: Superoxide
dismutase prevents increased lung vascular permeability after air emboli in
unanaesthetized sheep. Fed. Proc., 983: 405, 1981.
- Grogan J.B.: Altered neutrophil phagocytic function
in burn patients. J. Trauma, 16: 734-8, 1976.
- Hansbrough J.F., Field T.O., Gadd M.A., Soderberg
C.: Immune response modulation after burn injury: T-cells and antibodies. J. Bum Care
Rehabil., 8: 509-12,1987.
- Harlan J.M., Killen P.D., Harker L.A., Striker G.E.,
Wright D.G.: Neutrophil-mediated endothelial injury in vitro. J. Clin. Invest., 68:
1394-1403,1981.
- Henson P.: The immunologic release of constituents
from neutrophilic leukocytes. J. Inimunol., 107: 1535-46, 1974.
- Knaus W.A., Wagner D.P., Draper E.A., Zimmermann
J.E., Bergener M., Bastos P.G., Sirio C.A., Murphy D.J., Lotring T., Damiano A., Harrell
F.E.: The Apache III prognostic system. Risk prediction of hospital mortality for
critically ill hospitalized adults. Chest, 100: 1619-36, 1991.
- Messner R.P., Reed W.P., Palmer D.L.: A transient
defect in leukocytic bactericidal capacity. Clin. Immunol. Immunopathol., 1: 523-32, 1973.
- Messerschmidt 0., Langendorff H., Boraver H.:
Untersuchungen Ober Kombinationsschdden. St6rungen der Antik6rperbildung bei Mdusen, die
durch Verbrennungen und Ganzkbrperbestrahlungen belastet wurden. Strablentherapie, 139:
354, 1970.
- Ninnemann J.L.: Trauma, sepsis, and the immune
response. J. Bum Care Rehabil., 8: 462-67, 1987.
- O'Gormann R.B., Feliciano D.V., Mathews R., Mathews
B., Bitondo C.G., Mattox K.L., Jordan G.L.: Correlation of immunologic and nutritional
status with infectious complications after major abdominal trauma. Surgery, 99: 549-56,
1986.
- Pick E., Keisari Y.: A simple calorimetric method
for the measurement of hydrogen peroxide produced by cells in nature. J. Immunol. Methods,
38: 161-70, 1980.
- Rapaport F.T., Bachvaroff R.J.: Kinetics of humoral
responsiveness in severe thermal injury. Ann. Surg., 184: 51, 1976.
- Rittenbury M.S., Hanback L.D.: Phagocytic depression
in thermal injuries. J. Trauma, 7: 523-40,1967.
- Sacks T., Moldow C.F., Craddock P.R., Bowers T.K.,
Jacobs H.S.: Oxygen radicals mediate endothelial cell damage by complementstimulated
granulocytes. 1. Clin. Invest., 61: 1161-8, 1978.
- Sakai H., Daniels J.C., Beathard G.A., Lewis S.R.,
Lynch S.R., Ritzman S.E.: Mixed lymphocyte culture reaction in patients with acute thermal
bums. J. Trauma, 14: 53-7, 1974.
- Skornik W.A., Dressler D.P.: Lung bacteria]
clearance in the burnedrat. Ann. Surg., 172: 837-43, 1970.
- Solberg C.O., Hellum K.B.: Neutrophil granulocyte
function in bacterial infections. Lancet, 2: 727-30, 1972.
- Solomkin J.S., Nelson R.D., Chenowth D.E., Solem
L.D., Simmons R.L.: Regulation of neutrophil migratory function in burn injury by
complement activation products. nn. Surg., 200:742-6, 1984.
- Tate R.M., Repine J.E.: Neutrophils and the
respiratory distress syndrome. Am. Rev. Respir. Dis., 128: 552-9, 1983.
- Tschaikowsky K., Ruegheimer E.: Expression von
fMet-Leu-Phe Rezeptoren und Produktion von Sauerstoffradikalen in Granulozyten septischer
Patienten. Aenesthesist, 37: 140, 1988.
- Warden G.D., Mason A.D., Pruitt B.A.: Evaluation of
leukocyte chemotaxis in vitro in thermally injured patients. J. Clin. Invest., 54:
1001-4,1974.
- Weening R.S., Wever R., Loos D.: Quantitative
aspects of the production of superoxide radicals by phagocytizing human granulocytes. J.
Lab. Clin. Med., 85: 245-52,1975.
- Weiland J.E., Davies W.B., Holter U., Mohammed J.R.,
Dorinsky P.M., Gadek J.E.: Lung neutrophils in the adult respiratory distress syndrome.
Am. Rev. Respir. Dis., 133: 218-25,1986.
- Weissmann G., Smolen J.E., Korchak H.M.: Release of
inflammatory mediators from stimulated neutrophils. N. Engl. J. Med., 303: 27-34,1980.
- Westaby S.: Mechanisms of membrane damage and
surfactant depletion in acute lung injury. Intensive Care Med., 12: 2-5, 1986.
- Wood G.W., Volenee F.J., Mani M.M., Humphrey L.J.:
Dynamics of T-lymphocyte subpopulations and T-lymphocyte function following thermal
injury. Clin. Exp. Immunol., 31: 291-7, 197 8.
|



