Annals ofBurns and Fire Disasters - vol. IX -
n. 3 - September 1996
SOME
OBSERVATIONS ON MAST CELL NUMBERS IN STEROID-TREATED KELOIDS
Popchristova
E, * Mazgalova J.**
Department of
Pathology, Tzaritza loanna University Hospital, Sofia, Bulgaria Department of Tissue
Conservation, Pirogov Emergency Institute, Sofia
SUMMARY.
This study focuses on mast cells in steroid-treated keloids. Ten cases of untreated
and steroid-treated keloids were assessed for the number of mast cells. The use of light
microscopy morphometric analysis made it possible to establish a decreased number of mast
cells in steroid-treated keloids in comparison with that of mast cells in untreated skin
lesions. The reduction in the number of mast cells can be considered evidence for the
participation of these cells in the stages of keloid healing. The probable ways in which
mast cells and other connective tissue cells participate in the processes of steroid
keloidal treatment are discussed.
Mast cells, first described over 100 years ago, are well-examined cells of connective
tissue. They are located in the connective tissue throughout the body. Cutaneous mast
cells are normally present perivascularly in the dermis and subcutaneous tissue, with a
distribution ranging from 7000 to 20000 per rmul.' Mast cell granules release vasoactive
substances, chemotactic factors, enzymes, proteoglycans and smooth-muscle contracting
products .2 Mast cells are of bone-marrow origin' and are in close relationship with other
cells, particularly haematopoietic and mesenchymal cells.
Recent studies suggest that mast cells
participate in the pathogenesis of atopic and contact dermatitis, fibrosing reactions,
bullous pemphigoid, cutaneous disorders, neurofibromatosis, wound healing, polycythemia
vera, and psoriasis.' An increase in the number of mast cells was observed in localized
sclerodermia,' progressive systemic sclerosis,' and hypertrophic scars and keloids.1,1
Mast cell morphology was noted by Kischer and Bailey' in hypertrophic scars, mature scars
and normal skin, and, some years later, mast cell analysis was also performed during the
different stages of healing.' Data can be found in the literature regarding the number of
mast cells in pressure-therapy treated keloids,' but no data exist referring to the number
of mast cells in steroid-treated keloids.
The present study was conducted with a view to determining the number of mast cells in
materials from steroidtreated keloids. The aim of the investigation was thus to establish
mast cell numbers in skin lesions treated with steroids in comparison with mast cell
numbers in untreated keloids.
Materials and methods
A detailed description of the indications, methods of
cure, and morphological results of keloid treatment with steroids can be consulted in our
paper "Histomorphologic changes in keloids treated with KenacorC.
We used the method of light microscopy, combined with a morphometric analysis. Light
microscopic investigation was performed on materials from steroid-treated and untreated
keloids placed in 10% neutral paraformaldehyde and embedded in paraffin. Sections were cut
and stained with toluidine blue solution 1.5 x 10-'M in MacIlven buffer, and embedded with
Entellan for the histological identification of mast cells. The count of mast cells,
identified principally by their metachromasia, was performed until two months after
staining.
Both morphological and morphometric studies were carried out using a Laboval-type
microscope with net eyepiece and stage micrometer (Figs. 1,2) adapted to each other
according to the method of Awandilov.
The count of mast cells was performed at a
microscopic magnification of 40 x 6.3 x 1.6, where four divisions of the stage micrometer
correspond to three divisions of the net eyepiece. As one division of the stage micrometer
is equal to 0.01 min, the following measurements are 0.04 divided by 3 = 0.0133 min (one
division of the net eyepiece). The area of the smallest square in the eyepiece is thus
0.000177 rimil.
We observed 400 small squares in one field of vision of the microscope at this
magnification, i.e. the measuring area in a field of vision was 0.000177 mmI x 400 =
0.0708 min'.
The number of mast cells was established in 50 fields of vision for each slide, i.e. in an
area of 0.0708 irmil x 50 = 3.54 min'.
The data for the number of mast cells are presented as mean values for a 1 rum' area of
total dermis in untreated and steroid~treated keloids. The relation of the number of mast
cells in treated keloids to that in untreated keloids is presented as a percentage
proportion (Table 1). The results were subjected to statistical analysis and the
significance of P was determined.
The mast cell count was performed in five slides with untreated materials and five slides
with steroid-treated keloids.
 |
 |
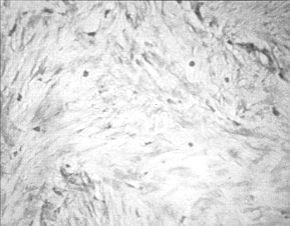 |
Fig. 3 - Histomorphological picture of mast cells in
untreated keloid. Toluidine blue stain. Original magnification 40 x. |
|
Results
Data from the mast cell count in total
dermis in steroid-treated and untreated keloids are presented in Table I. The mean
mast cell density of untreated keloids is significantly greater than that of
steroid-treated keloids.
The decreased number of mast cells in treated
keloids in comparison with the number in untreated lesions is presented as a percentage
relation (mean reduction 29.26%). Analysis of the data showed a significantly decreased
mast cell count in the dermis of treated keloids (P >0.001).
The classification of metachromatic stain intensity suggested that mast cells in untreated
keloids stained more intensely than those in treated keloids.
Metachromatic staining was very well expressed in some fields of untreated keloid tissue,
where the greatest number of mast cells was found. No metachromatic stain was observed in
the keloid tissue of steroid-treated keloids, where the metachromatic stain was located
only in mast cell granules.
The fields of vision where no mast cells were
seen represented 25-30% of the total number of fields of vision for each slide. The
highest number of six mast cells for one field of vision was seen in a slide with material
from untreated keloids.
In this study, only cells with clearly expressed metachromatic staining of the granules
were considered.
Discussion
In our research' on histomorphological changes in
steroid-treated keloids we gathered information about mast cells. We regarded our results
as evidence for the participation of mast cells in the processes of keloid healing. The
data reveal a reduction in the number of mast cells in total dermis in steroid-treated
keloids in comparison with untreated skin lesions. Our findings were consistent with the
results of Kischer et al.1,1 in their analysis of mast cells in hypertrophic scars. Once
again, some years after these detailed investigations, we have devoted our attention to
mast cells. By means of their biological mediators, mast cells participate actively in the
performance of all connective tissue processes. Data in the literature indicate the role
of fibroblasts and increased collagen synthesis in keloids The addition of hydrocortisone
or triamcinolone acetate to fibroblast cultures has been shown to retard the growth of
fibroblasts." It came to be known that the regulatory control mechanism that controls
the rate of collagen synthesis is sensitive to physiological levels of hydrocortisone.
| Type
of materials |
N |
Mast cell
density in 1 inni' |
p |
| |
1 |
13.55 |
|
| |
2 |
18.13 |
|
| Untreated
keloids |
3 |
13.04 |
|
| |
4 |
10.08 |
|
| |
5 |
12.34 |
|
| |
1 |
2.26 / 16.67% |
|
| |
2 |
3.02 / 16.65% |
|
| Treated
keloids |
3 |
4.20 / 32.20% |
0.01
>PA.00 1 |
| |
4 |
4.05 / 40.17% |
|
| |
5 |
5.04 / 40.61% |
|
|
| Table I - Mast cell density in total dermal zone of treated
and untreated keloids |
|
Oikarinen et al.11 suggested in 1978 that ster
oids act by binding on to the receptor sites in human fibroblasts. The addition of the
pharmacological agent antihistamine was found to be able to suppress the stimulation of
fibroblasts incubated with histamine." Patients with hypertrophic scars and keloids
have a significantly in creased tendency to allergy. The histamine content of keloid
tissue is higher than that of normal scar tissue" and is related to the increased
number of mast cells there. This suggests that the enhanced mast cell activity or count
may be a common denominator that predisposes such persons both to allergy and to abnormal
wound healing. 17 The large number of biologically active factors released from mast cell
granules may stimulate the increased collagen synthesis and fibroblast
proliferation."," Fibroblasts themselves have been shown to influence mast cell
differentiation and granule synthesis."," Fine regulatory interactions have been
shown between the two main types of connective tissue cells, fibroblasts and mast cells.
Electron and transmission electron microscopy examinations of fibroblast/mast cell
co-cultures suggest that mast cells are capable of extending granule-laden pseudopods to
the surface of opposing cells." These mechanisms of transgranulation and
degranulation are probably the way in which mast cells expose constituents of their
granules to surrounding cells.
Keloid tissue enriched with biologically
active products may be accepted as a condition in vivo in which the abnormal histamine
content stimulates fibroblast proliferation and increased collagen synthesis.
Non-immunological stimuli and/or pharmacological agents might provoke an inhibition of
this stimulation and decrease the fibroblast function. There are data in the literature
that consider the mechanisms by which a cortisone derivate may depress collagen synthesis.
This probably occurred also in our investigation. The way steroids act on mast cells and
lead to their decreased number remains unclear to us. We accept that some mast cells might
become indistinguishable from surrounding cells as a result of cellular degranulation,
during microscopic analysis. Our electron microscopy observations, although devoid of
morphological data demonstrate a single mast cell in the dermal zone oi treated keloids.
The main feature of our study is the great reduction in the number of mast cells in
steroid-treated keloids. The uniform distribution of mast cells in the total dermis is
evidence of our correct clinical treatment. We consider the reduced mast cell count to be
an indication of the participation of mast cells not only in the formation of keloids but
also in the successive stages of their treatment.
RESUME. Dans cette
�tude les Auteurs consid�rent la question des mastocytes pr�sents dans les ch�lo�des
trait�es avec les st�ro�des. Dix cas de ch�lo�des non trait�es et trait�es avec les
st�ro�des ont �t� �valu�s pour le num�ro de mastocytes. Avec l'emploi de l'analyse
morphom�trique au microscope optique ils ont observ� un num�ro r�duit de mastocytes
dans les ch�lo�des trait�es avec les st�ro�des par rapport au num�ro de mastocytes
dans les l�sions cutan�es non trait�es. La r�duction dans le num�ro des mastocytes
est interpr�t�e comme �vidence pour la participation de ces cellules dans les phases de
la gu�rison des ch�lo�des. Les auteurs discutent enfin le r�le probable des mastocytes
et des autres cellules du tissu conjonctif dans les processus du traitement des
ch�lo�des avec les st�ro�des.
BIBLIOGRAPHY
- Mikhail G.R., Miller-Milinska A.: Mast cell
population in human skin. J. Invest. Dermatol., 43: 249-54, 1964.
- Kerdel EA., Soter N.A.: The mast cell in
mastocytosis and pediatric dermatologic disease. Adv. Dermatol., 4: 159-82, 1989.
- Hatanaka K., Kitamura Y., Nishimune Y.: Local
development of mast cell from bone-marrow-derived precursors in the skin of mice. Blood,
53: 142-7, 1979.
- Rothe M., Nowak M., Kerdel E: The mast cell in
health and disease. J. Amer. Acad. Dermatol., 23: 615-24, 1990.
- Nishioka K., Kobayashi Y, Katayama J. et al.: Mast
cell numbers in diffuse sclerodermia. Arch. Dermatol., 123: 205-8, 1987.
- Hawkins R.A., Claman H.N., Clark
R.A.F. et al.: Increased dermal mast cell populations in progressive systemic sclerosis: A
link in chronic fibrosis. Ann. Intern. Med., 102: 182-6, 1985.
- Kiscber C.W., Bailey J.F.: The mast
cell in hypertrophic scars. Texas Rep. Biol. Med., 30: 327-39, 1972.
- Kischer C.W., Bunce H., Shetlar MR.:
Mast cell analysis in hypertrophic scars, hypertrophic scars treated with pressure and
mature scars. J. Invest. Dermatol., 70: 355-7, 1978.
Boyadjiev C., Popchristova
E., Mazgalova J.: Histomorphologic changes in keloids treated with Kenacort. J. Trauma,
Injury, Infection and Critical Care, 38: 299-302, 1995.
Avtandilov G.G.: "Meditzinskaya
morphometria", Meditzina, Moscow, 1990.
Murray J.C., Pollack SN., Pinnell
S.R.: Keloids: A review. J. Am. Acad. Dermatol., 4: 461-70, 1981.
Datubo-Brown D.D.: Keloids: A review
of the literature. Br. J. Plast. Surg., 43: 70-7, 1990.
McCoy B.J., Diegelmann R.F., Cohen
I.K.: In vitro inhibition of cell growth, collagen synthesis and prolyl hydrolase activity
by triamcinolone acetonide. Proc. Soc. Exp. Biol. Med., 163: 216-22, 1980.
Oikarinen A., Oikarinen H.,
Meeker C.A. et al.: Glucocorticoid receptor in cultured human skin fibroblasts: evidence
for down regulation of receptor by glucocorticoid hormone. Acta Dermato-Venerol
(Stockholm), 67: 461, 1987.
Topol B.M., Lewis VL., Benveniste
K.: The use of antihistamine to retard the growth of fibroblasts derived from human skin,
scar and keloid. Plast. Reconstr. Surg., 68: 227, 1981.
Cohen I.K., Beaven M.A., Horakova Z.
et al.: Histamine and collagen synthesis in keloids and hypertrophic scar. Surg. Forum,
23:509-10, 1972.
Smith C.J., Smith J.C., Finn M.C.:
The possible role of mast cells (allergy) in the production ofkeloid and hypertrophic
scarring. J.Bum Care Rehabil., 8: 126-31, 1987.
Russel J.D., Russel S.B., Trupin
K.M: The effect of histamine on the growth of cultured fibroblasts isolated from normal
and keloid tissue. J. Cell. Physiol., 93: 389-94, 1977.
Hatamochi A., Fujiwara K., Ueki H.:
Effects of histamine on collagen synthesis by cultured fibroblasts derived from guinea pig
skin. Arch. Dermatol. Res., 277: 60-4, 1985.
Levi-Schaffer F., Austen K.F.,
Gravallese P.M., et al.: Coculture of interleukin-3 -dependent mouse mast cells with
fibroblasts results in phenotypic change of the mast cells. Proc. Nad. Acad. Sci. USA,
83:6485-8, 1986.
Davidson S., Mansour A., Gallily R.
et al.: Mast cell differentiation depends on T cells and granule synthesis on
fibroblasts.Immunology, 48: 439-52, 1983.
Greenberg G., Bumstock G.: A novel
cell-to-cell interaction between mast cells and other cell types. Exp. Cell. Res., 147:
1-13, 1983.
| This paper was
received on 8 March 1996. Address
correspondence to: Prof. Janet Mazgalova, Ph.D., Department of Tissue Conservation,
Pirogov Emergency Institute, 21 Makedoniya Str., 1606 Sofia, Bulgaria. |
|


